Executive Summary
As part of the MDR or IVDR conformity assessment and ongoing audit cycle, manufacturers are subject to regular surveillance activities conducted by their Notified Body. This audit cycle includes Unannounced Audit Visits (UAVs), carried out at the premises of the manufacturer or suppliers and/or key subcontractors without prior notice.
Unannounced Audit Visits are intended to verify that day-to-day operational practices consistently reflect the approved technical documentation and the quality management system (QMS) supporting the certified device.
UAVs are a critical assessment activity for ensuring transparency and demonstrating compliant processes are consistently applied. UAVs confirm the robustness of routine operations and demonstrate the manufacturer’s ongoing commitment to MDR or IVDR compliance.
Unannounced Audit Opening
An Unannounced Audit Visit (UAV) is conducted by a team typically consisting of a QMS assessor and a Technical Specialist. The audit formally begins the moment the team arrives on the manufacturer’s site.
Upon arrival, the Notified Body audit team presents its credentials, explains the purpose of the visit, and requests immediate access to operational areas. This approach is intentional: the objective of a UAV is to observe the organization as it truly operates under normal, day-to-day conditions, without advance preparation or staging.
To support this objective, the opening meeting is kept brief and focused. Prolonged introductions or presentations are avoided so the audit team can move directly into active operational areas, where real processes, decisions, and controls are taking place.
Manufacturers holding CE certification are expected to be familiar with this regulatory requirement under the MDR and IVDR. Effective preparation is demonstrated through prompt cooperation, clear internal communication, and the ability to facilitate access without disruption. This readiness not only enables an efficient audit but also reflects the transparency, control, and compliance expected within the EU regulatory framework.
How a UAV Unfolds and What the Assessment Team Focuses on
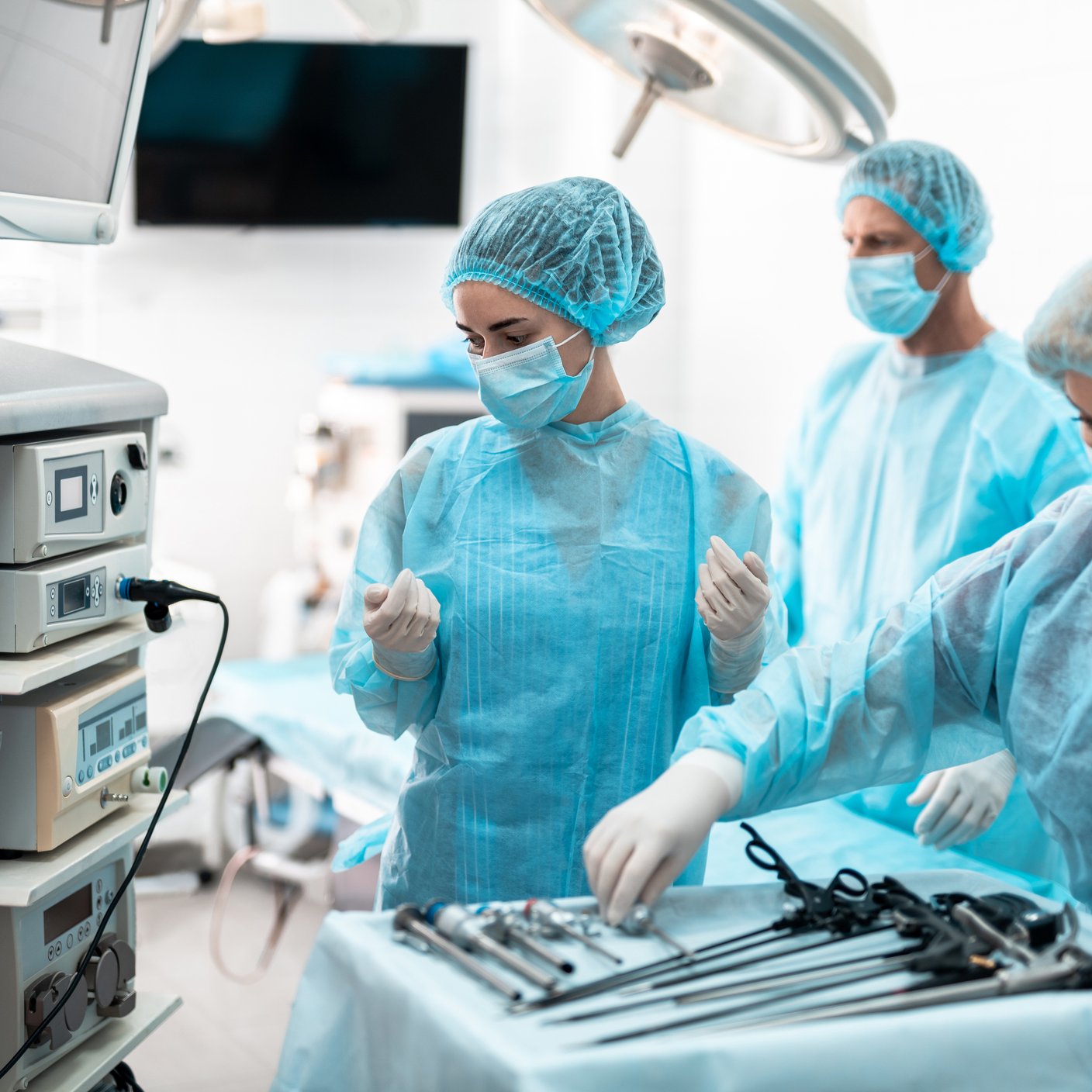
Once the UAV team enters the operational environment, the audit proceeds according to a structured plan. A UAV typically covers a full working day and focuses on verifying how the manufacturer operates in practice. The assessment team directly compares day-to-day activities with the requirements defined in the QMS and technical documentation. This includes:
- Real-time observation of manufacturing or service activities.
- Review of records generated during routine operations.
- Discussions with personnel performing the work.
- Verification that actual outputs match documented procedures and regulatory requirements.
A UAV team combines quality system and technical expertise to enable an efficient and well-balanced assessment. During these visits, the Notified Body audit team may encounter a range of operational situations, such as overlapping audits, temporary absence of key personnel, or internally scheduled activities.
Manufacturers should be prepared to adjust quickly in these circumstances to ensure that relevant personnel, processes, and records remain accessible for assessment. Clear internal coordination and contingency planning help minimize disruption and support a smooth audit flow.
UAVs may also coincide with periods of reduced activity or limited staffing. For this reason, certificate holders are required to inform the Notified Body in advance of planned shutdowns, holidays, or significant operational changes. Providing timely and accurate information allows the UAV to be scheduled at an appropriate time, ensures appropriate assessment of ongoing operations, and helps avoid unnecessary delays or inefficiencies.
The Outcome of a UAV and the Value it Brings
The UAV concludes with the audit team producing a clear summary of observations and a recommendation on the continuation of certification. Where nonconformities are identified, the certificate holder receives specific requirements and defined timelines for corrective action.
Continuous readiness reinforces adherence to established processes and aligns day-to-day operations more closely with regulatory requirements. This operational discipline supports consistent performance, reduces audit findings, and strengthens the overall QMS.
Although unannounced audits may place temporary demands on resources and scheduling, they provide direct evidence of process control under day-to-day operating conditions. Manufacturers that approach UAVs in a structured and transparent manner are well positioned to demonstrate sustained compliance, product quality, and patient safety.
A well-managed UAV offers objective confirmation that the quality management system is effective in practice - not only on paper.
Key Takeaways
- Immediate access and readiness: UAVs start as soon as the audit team arrives on your site. Ensure personnel, records, and operational areas are accessible without delay.
- Audit focus: The team observes day-to-day operations, reviews records, speaks with staff, and verifies that outputs match QMS procedures and regulatory requirements.
- Maintain continuous preparedness: Ongoing readiness strengthens daily processes and aligns operations with MDR/IVDR expectations.
- Plan for operational variability: Notify the Notified Body in advance of shutdowns, holidays, or major disruptions. Internal coordination ensures key personnel and processes remain available.
- Actionable outcomes: UAVs confirm if the QMS works effectively in practice. Nonconformities are communicated clearly with defined corrective action timelines.
- Value of UAVs: Approached proactively, UAVs demonstrate process robustness, regulatory compliance, product quality, and patient safety, offering objective validation of your QMS.
Author
Laura De Francesco, Service Delivery Manager
Regulatory Services, BSI